Files
Submission Date
Fall 2021
Abstract
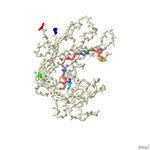
Marine bacterial laminarinase cuts polysaccharides in the process of remineralization, a key process in the ocean biogeochemical cycling of nutrients. We reviewed the protein model of 6JH5 in Jmol to highlight the important areas of the model that relate to thermostability and function and the effects of global climate change on the protein. The catalytic cleft of Glu135 and Glu140 was vital to the depolymerization mechanism. Substrate chain positions 130-143, specifically Trp130 being used for recognition, were important to the proficiency of the structure. The calcium ion on the opposite side of the β-sheet from catalytic cleft increased its degrading activity and thermostability. The residue interactions with Glc(−1) and Glc(−2) were unveiled to be crucial for β-1,3-glycosidic bond selectivity by the enzyme. Previous studies also showed the residue interactions were also important to the protein’s thermostability and thermophilicity. Our research found that the protein’s function will be negatively impacted by global climate changes as temperature beyond the protein’s optimal temperature (6JH5: 20°C) caused by climate change will also decline activity as hydrogen bonds between proteins weaken and being to denature. The function of our protein is important in the biogeochemical cycling of nutrients in the ocean and without its high activity, the cycling of nutrients and DOM like carbon and nitrogen won’t be as proficient.
Recommended Citation
Hellenbrand, Heidi; Harris, Rachel; and Villanueva, Chino, "Highlighting How the Structure of Marine Bacterial Laminarinase Can Improve Biogeochemical Cycling During Global Climate Change" (2021). Protein Modeling Reports. 1.
https://nsuworks.nova.edu/protein_modeling_reports/1
Poster
Bacterial Laminarinase Presentation ‐ Made with Clipchamp.mp4 (21466 kB)
Presentation Video
FINAL ppt presentation.pdf (9460 kB)
Presentation